HO CHI MINH CITY USES MOLNUPIRAVIR FOR TREATING COVID-19 PATIENTS AT HOME
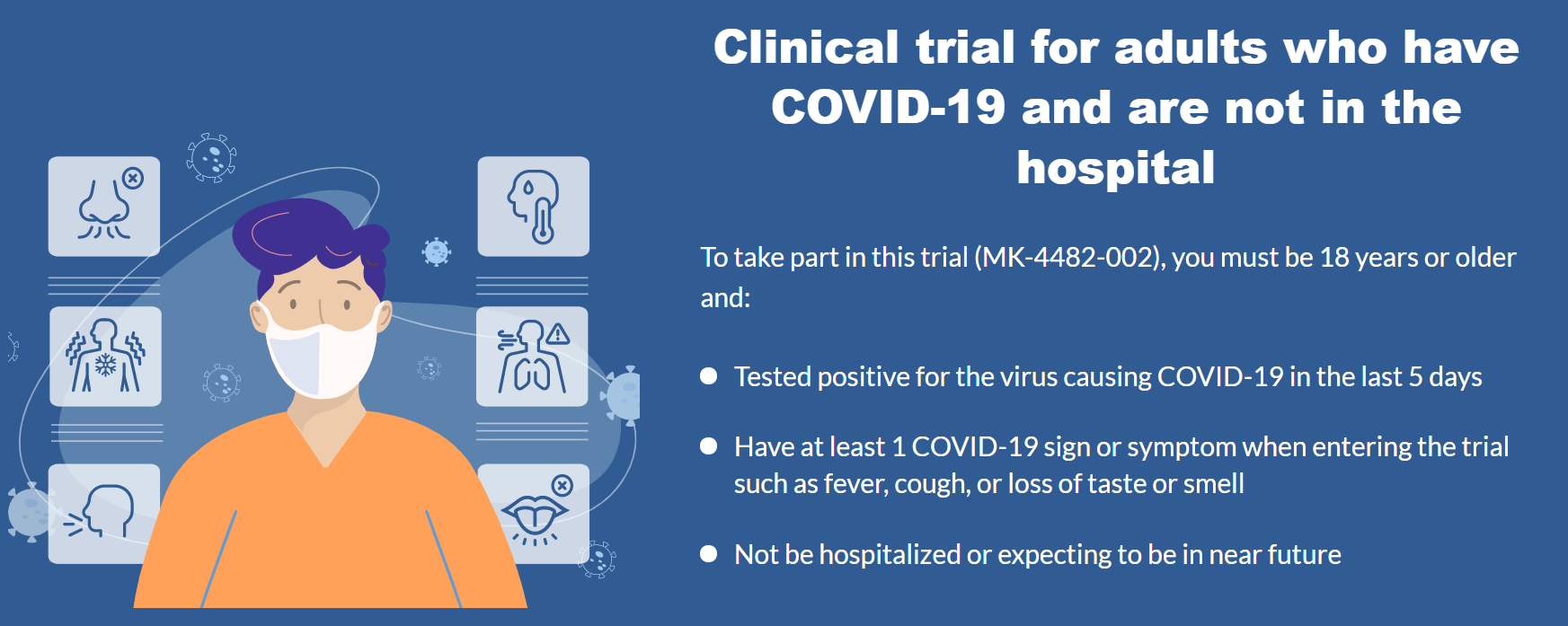
According to Minister of Health Nguyen Thanh Long, the Ministry of Health and the Department of Health of Ho Chi Minh City start using the experimental antiviral drug Molnupiravir to treat Covid-19 patients with mild-symptom from August 27, 2021. He said this while inspecting the Covid-19 prevention and control task in Ho Chi Minh City’s